Pages
Thursday, 31 July 2008
MicroRNAs Fine-tune Protein Synthesis
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Monday, 28 July 2008
More Trouble Ignite for Bush’s ESC Lines
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Friday, 25 July 2008
Stem Cell Forum in China Demonstrate Cutting Edge Research
 At the Forum, which was chaired by Dr. Sean Hu, Chairman of Beike Biotechnology Co., Ltd., some of the world's most respected researchers presented their latest laboratory findings and clinical trial results in using stem cells to treat common ailments like heart disease and nervous system diseases such as cerebral palsy, spinal cord injury, muscular dystrophy, and optic nerve hypoplasia (ONH). Biologists attending the Mandarin Chinese language Forum came from leading research centers in nearly all of China's major cities, and as far away as the United States, Canada, India, Australia, and Malaysia.
A special symposium entitled Advanced iPS Cell Technology, convened as part of the Forum, brought together for the first time over a dozen scientists involved in cutting edge stem cell research at some of America and China's most respected science universities and research labs. A potential substitute for embryonic stem cells, genetically programmable iPS cells are widely believed to be at the very forefront of the field's most exciting research frontier as iPS cells theoretically could deliver the greatest therapeutic benefit. Since iPS cells can be produced from the building blocks in any individual's own body, they bring the potential that people may someday be able to heal a range of sicknesses from tissue cells they have banked themselves.
At the symposium, renowned stem cell scientists like Dr. Hu Jifan of the Palo Alto VA Health Care System under the Stanford University Medical School, Dr. Xiang Peng of Sun Yat Sen University, Dr. Zhang Yaou of Tsing Hua University, Dr. Kong Hsiang-Fu of the Chinese University of Hong Kong, Dr. Zhou Xiangjun of Shanghai Jiaotong University, and Dr. Yong-Jian Geng of the University of Texas Health Science Center at Houston and the Texas Heart Institute, discussed their own latest advancements since recent breakthroughs at the University of Wisconsin and Kyoto University in Japan. The scientists also discussed mutual cooperation and research oversight opportunities, creation of ethical research guidelines in China, and future iPS cell research directions.
At the symposium, a special subgroup of China's leading cardiovascular scientists like Dr. Chen Haozhu of Fudan University, Dr. Wang Shiwen of the Chinese PLA General Hospital, and Dr. Zhang Fumin of Nanjing Medical University conferred on the use of stem cells in heart disease treatment, as well as new research methods and ethical responsibilities in that field.
Dr. Yong-Jian Geng, M.D., Ph.D, Director of the Heart Failure and Stem Cell Research Lab at the Texas Heart Institute and Professor of Medicine at the University of Texas Medical School in Houston, said:
"Scientists and clinicians have been doing incredible research on stem cells, and generating many exciting results. I feel confident more breakthroughs will be coming soon. I was very pleased and honoured to be invited to participate in this discussion, and I am willing to help Chinese investigators establish guidelines that will, in my opinion, help develop a global standard for the stem cell therapies."
Dr. Hu Jifan, senior research scientist at the Palo Alto VA Health Care System under the Stanford University Medical School said:
"Leading scientists in China are constantly finding ways to create more effective iPS cells. Through this forum we agreed to help verify each other's work, share experiences, and ensure safe, high-standard research takes place so that we may someday create the conditions for effective clinical treatments and industrial scale production of iPS cells. While no one knows yet when these conditions might be met, this Forum certainly helped speed up the process by bringing together some of the world's best minds working towards these goals."
Presentations at the 2008 China Stem Cell Technology Forum covered a wide range of new biological advancements as Chinese scientists hone in on the global goal of bringing practical stem cell treatments to market. Other topics covered included findings on the most potent combinations of stem cells, effective cell processing methods, the latest clinical trials targeting diseases like heart conditions, muscular dystrophy and optic nerve hypoplasia (ONH), and more yet to be published stem cell research.
At the Forum, which was chaired by Dr. Sean Hu, Chairman of Beike Biotechnology Co., Ltd., some of the world's most respected researchers presented their latest laboratory findings and clinical trial results in using stem cells to treat common ailments like heart disease and nervous system diseases such as cerebral palsy, spinal cord injury, muscular dystrophy, and optic nerve hypoplasia (ONH). Biologists attending the Mandarin Chinese language Forum came from leading research centers in nearly all of China's major cities, and as far away as the United States, Canada, India, Australia, and Malaysia.
A special symposium entitled Advanced iPS Cell Technology, convened as part of the Forum, brought together for the first time over a dozen scientists involved in cutting edge stem cell research at some of America and China's most respected science universities and research labs. A potential substitute for embryonic stem cells, genetically programmable iPS cells are widely believed to be at the very forefront of the field's most exciting research frontier as iPS cells theoretically could deliver the greatest therapeutic benefit. Since iPS cells can be produced from the building blocks in any individual's own body, they bring the potential that people may someday be able to heal a range of sicknesses from tissue cells they have banked themselves.
At the symposium, renowned stem cell scientists like Dr. Hu Jifan of the Palo Alto VA Health Care System under the Stanford University Medical School, Dr. Xiang Peng of Sun Yat Sen University, Dr. Zhang Yaou of Tsing Hua University, Dr. Kong Hsiang-Fu of the Chinese University of Hong Kong, Dr. Zhou Xiangjun of Shanghai Jiaotong University, and Dr. Yong-Jian Geng of the University of Texas Health Science Center at Houston and the Texas Heart Institute, discussed their own latest advancements since recent breakthroughs at the University of Wisconsin and Kyoto University in Japan. The scientists also discussed mutual cooperation and research oversight opportunities, creation of ethical research guidelines in China, and future iPS cell research directions.
At the symposium, a special subgroup of China's leading cardiovascular scientists like Dr. Chen Haozhu of Fudan University, Dr. Wang Shiwen of the Chinese PLA General Hospital, and Dr. Zhang Fumin of Nanjing Medical University conferred on the use of stem cells in heart disease treatment, as well as new research methods and ethical responsibilities in that field.
Dr. Yong-Jian Geng, M.D., Ph.D, Director of the Heart Failure and Stem Cell Research Lab at the Texas Heart Institute and Professor of Medicine at the University of Texas Medical School in Houston, said:
"Scientists and clinicians have been doing incredible research on stem cells, and generating many exciting results. I feel confident more breakthroughs will be coming soon. I was very pleased and honoured to be invited to participate in this discussion, and I am willing to help Chinese investigators establish guidelines that will, in my opinion, help develop a global standard for the stem cell therapies."
Dr. Hu Jifan, senior research scientist at the Palo Alto VA Health Care System under the Stanford University Medical School said:
"Leading scientists in China are constantly finding ways to create more effective iPS cells. Through this forum we agreed to help verify each other's work, share experiences, and ensure safe, high-standard research takes place so that we may someday create the conditions for effective clinical treatments and industrial scale production of iPS cells. While no one knows yet when these conditions might be met, this Forum certainly helped speed up the process by bringing together some of the world's best minds working towards these goals."
Presentations at the 2008 China Stem Cell Technology Forum covered a wide range of new biological advancements as Chinese scientists hone in on the global goal of bringing practical stem cell treatments to market. Other topics covered included findings on the most potent combinations of stem cells, effective cell processing methods, the latest clinical trials targeting diseases like heart conditions, muscular dystrophy and optic nerve hypoplasia (ONH), and more yet to be published stem cell research.
 Beike Biotechnology Co., Ltd., hosted the event along with the China National Center for Biotechnology Development (CNCBD) under China's Ministry of Science and Technology (MOST), the Jiangsu Provincial Department for Science and Technology, and the Taizhou Government. The 2008 China Stem Cell Technology Forum was attended by MOST officials, the Vice Governor of Jiangsu Province Mr. Zhang Taolin, and Taizhou Mayor Mr. Yao Jianhua.
Dr. Sean Hu, the Chairman of Beike Biotech said:
"Chinese science, and indeed global stem cell research, achieved a great deal through this forum. Beike is proud to work with China to bring together so many globally known Mandarin speaking stem cell scientists to better communicate and share resources regarding what may very well become the future of healthcare."
About the China Medical City (CMC):
Jiangsu Province is considered the number one location for China's medical industry based on revenue generated over the past 5 years. The city of Taizhou in Jiangsu is not only the hometown of China's President Hu Jintao, but is considered the fastest growing medical industry location in Jiangsu, with over 35 % annual growth in that time. Established by the Chinese Government in 2005 and consisting of 20-25 square kilometres in the heart of Taizhou City, China Medical City (CMC) is fully supported by China's local and national governments. CMC is emerging as a strong leader in China's efforts to develop a streamlined pharmaceutical and medical materials industry that concentrates all medical services and support in one location. Businesses located in CMC carry out a range of manufacturing and support services including research and development, creation and processing of medical materials, distribution, comprehensive healthcare delivery solutions, and patent filing support.
About Beike Biotechnology Company Limited:
Beike is a biotechnology company that was founded in July 2005 with capital from Beijing University, Hong Kong University of Science and Technology and Shenzhen City Hall when it commercialized stem cell technology that had been in research since 1999. The research and clinical work comes from collaborations with leading institutions in China including of Tsinghua University, Beijing University, Hong Kong University of Science and Technology, No. 3 Army Medical University, Zhongshan Medical University, Guiyang Medical College and Zhengzhou University. Over 250 patients every month are treated with Beike's stem cells in leading hospitals throughout China. Patient experiences from treatments can be found at Stem Cell China News.
SOURCE: Adopted from material from Beike Biotechnology Company Limited.
See also:
Beike Biotech Opens Comprehensive Stem Cell Storage and Processing Facility in China
Medical News Today - 20 June 2008
.........
ZenMaster
Beike Biotechnology Co., Ltd., hosted the event along with the China National Center for Biotechnology Development (CNCBD) under China's Ministry of Science and Technology (MOST), the Jiangsu Provincial Department for Science and Technology, and the Taizhou Government. The 2008 China Stem Cell Technology Forum was attended by MOST officials, the Vice Governor of Jiangsu Province Mr. Zhang Taolin, and Taizhou Mayor Mr. Yao Jianhua.
Dr. Sean Hu, the Chairman of Beike Biotech said:
"Chinese science, and indeed global stem cell research, achieved a great deal through this forum. Beike is proud to work with China to bring together so many globally known Mandarin speaking stem cell scientists to better communicate and share resources regarding what may very well become the future of healthcare."
About the China Medical City (CMC):
Jiangsu Province is considered the number one location for China's medical industry based on revenue generated over the past 5 years. The city of Taizhou in Jiangsu is not only the hometown of China's President Hu Jintao, but is considered the fastest growing medical industry location in Jiangsu, with over 35 % annual growth in that time. Established by the Chinese Government in 2005 and consisting of 20-25 square kilometres in the heart of Taizhou City, China Medical City (CMC) is fully supported by China's local and national governments. CMC is emerging as a strong leader in China's efforts to develop a streamlined pharmaceutical and medical materials industry that concentrates all medical services and support in one location. Businesses located in CMC carry out a range of manufacturing and support services including research and development, creation and processing of medical materials, distribution, comprehensive healthcare delivery solutions, and patent filing support.
About Beike Biotechnology Company Limited:
Beike is a biotechnology company that was founded in July 2005 with capital from Beijing University, Hong Kong University of Science and Technology and Shenzhen City Hall when it commercialized stem cell technology that had been in research since 1999. The research and clinical work comes from collaborations with leading institutions in China including of Tsinghua University, Beijing University, Hong Kong University of Science and Technology, No. 3 Army Medical University, Zhongshan Medical University, Guiyang Medical College and Zhengzhou University. Over 250 patients every month are treated with Beike's stem cells in leading hospitals throughout China. Patient experiences from treatments can be found at Stem Cell China News.
SOURCE: Adopted from material from Beike Biotechnology Company Limited.
See also:
Beike Biotech Opens Comprehensive Stem Cell Storage and Processing Facility in China
Medical News Today - 20 June 2008
.........
ZenMaster For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Thursday, 24 July 2008
Adult Stem Cells Activated in Mammalian Brain
First, the UCI researchers sought to determine the true location of stem cells in the rats by looking for polarized cells, which have different sets of proteins on opposite sides so that when one divides it can produce two different products. Polarization gives rise to asymmetric cell division, which produces one copy of the parent and a second cell that is programmed to turn into another cell type. Asymmetric cell division is the defining characteristic of a stem cell. On rat brain samples, the researchers applied antibodies to identify proteins that may be involved in asymmetric cell division, and they found that polarization exists on the ependymal cells. "It couldn't have been a stronger signal or clearer message. We could see that the only cells undergoing asymmetric cell division were the ependymal cells," Gleason said. Next, they gave a drug to induce cell division in the rats and examined their brains at intervals ranging from one to 28 days after the treatment. At each interval, they counted cells that were dividing in the ependymal layer. They found the most division at 28 days, when about one-quarter of the ependymal cells were dividing. Previous studies by researchers at other institutions were successful in getting only a few cells to divide in that layer. "One interpretation of previous studies is there are scattered stem cells in the ependymal layer, and it is hard to locate them," Bryant said. "But we believe that all of the ependymal cells are stem cells, and that they all have the ability to be activated." Researchers don't know yet what sparks cell division at the molecular level, but learning that process and how to control it could lead to a safe, effective stem cell therapy. About the University of California, Irvine: The University of California, Irvine is a top-ranked university dedicated to research, scholarship and community service. Founded in 1965, UCI is among the fastest-growing University of California campuses, with more than 27,000 undergraduate and graduate students and nearly 2,000 faculty members. The third-largest employer in dynamic Orange County, UCI contributes an annual economic impact of $3.6 billion. See also: Adult Stem Cells Reprogrammed in the Brain CellNEWS - Monday, 30 June 2008 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
India to Set Up Stem Cell Research Unit in Bangalore
 India's Union Cabinet Thursday gave its approval for the establishment of an Institute for Stem Cell Science and Regenerative Medicine (SCSRM) in Bangalore.
The institute will help in “developing policies relating to stem cell technologies, identify needs, facilitate conceptualisation and design of new technologies,” Priya Ranjan Dasmunsi, Information and Broadcasting Minister, told reporters.
The SCSRM institute would be an autonomous institution of the Department of Biotechnology and would be set up at a total cost of Rs. 203.10 crores (approx. US$ 48.4M).
The cabinet also approved the integration of Stem Cell Centre (SCC) at the Christian Medical College, Vellore, now funded by the department of biotechnology, with the Bangalore institute.
Source: Adopted from Press Release from the Press Information Bureau, Government of India.
.........
ZenMaster
India's Union Cabinet Thursday gave its approval for the establishment of an Institute for Stem Cell Science and Regenerative Medicine (SCSRM) in Bangalore.
The institute will help in “developing policies relating to stem cell technologies, identify needs, facilitate conceptualisation and design of new technologies,” Priya Ranjan Dasmunsi, Information and Broadcasting Minister, told reporters.
The SCSRM institute would be an autonomous institution of the Department of Biotechnology and would be set up at a total cost of Rs. 203.10 crores (approx. US$ 48.4M).
The cabinet also approved the integration of Stem Cell Centre (SCC) at the Christian Medical College, Vellore, now funded by the department of biotechnology, with the Bangalore institute.
Source: Adopted from Press Release from the Press Information Bureau, Government of India.
.........
ZenMaster For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Omega-3 Fatty Acids Could Slow Acute Wound Healing
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Your Lower Belly Fat Is Rich in Stem Cells
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Lick Your Wounds!!!
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Tuesday, 22 July 2008
Spinal Cord Stem Cells May Act as Nerve Repair System
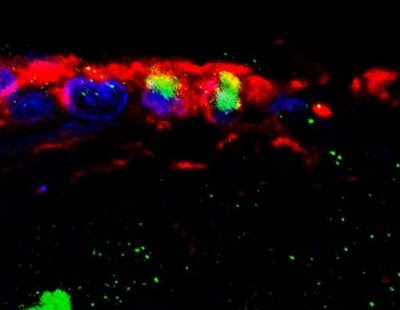
 Ependymal cells in the adult spinal cord self-renew in vivo. Stack of 14 images depicting vimentin expressing ependymal cells (purple) of the central canal that have incorporated BrdU (have divided, orange). Spinal cord neurons are labelled with NeuN (white). Credit: Image/Design by Marie Carlén; confocal image by Fanie Barnabé-Heider.
Ependymal cells in the adult spinal cord self-renew in vivo. Stack of 14 images depicting vimentin expressing ependymal cells (purple) of the central canal that have incorporated BrdU (have divided, orange). Spinal cord neurons are labelled with NeuN (white). Credit: Image/Design by Marie Carlén; confocal image by Fanie Barnabé-Heider.
The study uncovers the molecular mechanism underlying the tantalizing results of the rodent and primate and goes one step further: By identifying for the first time where this subpopulation of cells is found, they pave a path toward manipulating them with drugs to boost their inborn ability to repair damaged nerve cells. "The ependymal cells' ability to turn into several different cell types upon injury makes them very interesting from an intervention aspect: Imagine if we could regulate the behaviour of this stem cell population to repair damaged nerve cells," Meletis said. Upon injury, ependymal cells proliferate and migrate to the injured area, producing a mass of scar-forming cells, plus fewer cells called oligodendrocytes. The oligodendrocytes restore the myelin, or coating, on nerve cells' long, slender, electrical impulse-carrying projections called axons. Myelin is like the layer of plastic insulation on an electrical wire; without it, nerve cells don't function properly. "The limited functional recovery typically associated with central nervous system injuries is in part due to the failure of severed axons to regrow and reconnect with their target cells in the peripheral nervous system that extends to our arms, hands, legs and feet," Meletis said. "The function of axons that remain intact after injury in humans is often compromised without insulating sheaths of myelin." If scientists could genetically manipulate ependymal cells to produce more myelin and less scar tissue after a spinal cord injury, they could potentially avoid or reverse many of the debilitating effects of this type of injury, the researchers said. This study was supported by grants from the Swedish Research Council, the Swedish Cancer Society, the Foundation for Strategic Research, the Karolinska Institute, EuroStemCell and the Christopher and Dana Reeve Foundation. Reference: Spinal Cord Injury Reveals Multilineage Differentiation of Ependymal Cells Konstantinos Meletis, Fanie Barnabé-Heider, Marie Carlén, Emma Evergren, Nikolay Tomilin, Oleg Shupliakov, Jonas Frisén PLoS Biol 6(7): e182 doi:10.1371/journal.pbio.0060182 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Sunday, 20 July 2008
The Genetics of the White Horse Unravelled
DNA from more than 900 horses was analysed, several of them from the white Lipizzaner horses from the Spanish Riding School in Vienna. In all white horses, but not in any black, brown or chestnut coloured horse, the exact same DNA change was found, a large segment of DNA duplication. This mutation does not change any protein coding gene, but regulate how active two other genes will be. The Grey horse is also very interesting from a medical point of view since the mutation also predisposes for development of melanoma. About 75% of Grey horses older than 15 years of age have a benign form of melanoma that in some cases develops into a malignant melanoma. Thus, the study reported today has also given new insight in a molecular pathway that may lead to tumour development. “We propose that the Grey mutation stimulates growth of melanocytes and that this leads to a premature loss of the melanocyte stem cells needed for hair pigmentation whereas the mutation promotes an expansion of some of the melanocytes causing skin pigmentation,” says Leif Andersson. Domestic animals constitute extraordinary models for evolution of biological diversity as already recognized by Charles Darwin. The white horse is a beautiful illustration of the importance of regulatory mutations as a major underlying mechanism for phenotypic diversity within and between species. The Grey mutation does not change any protein structure but it affects the genetic regulation of two genes. The researchers found that the white horses carry an extra copy of a DNA segment located in one of these genes. “It is very likely that regulatory mutations like the one we found in these white horses constitute the dominating class of mutations explaining differences between breeds of domestic animals as well as between species like humans and chimpanzee,” concludes Leif Andersson. Referens: A cis-acting regulatory mutation causes premature hair greying and susceptibility to melanoma in the horse Gerli Rosengren Pielberg, Anna Golovko, Elisabeth Sundström, Ino Curik, Johan Lennartsson, Monika H Seltenhammer, Thomas Druml, Matthew Binns, Carolyn Fitzsimmons, Gabriella Lindgren, Kaj Sandberg, Roswitha Baumung, Monika Vetterlein, Sara Strömberg, Manfred Grabherr, Claire Wade, Kerstin Lindblad-Toh, Fredrik Pontén, Carl-Henrik Heldin, Johann Sölkner & Leif Andersson Nature Genetics Published online: 20 July 2008, doi:10.1038/ng.185 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Friday, 18 July 2008
Standards in Stem Cell Research Needed
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Sunday, 13 July 2008
Stem Cell Tourism Seen as ‘Medical Hoax’ by Specialists
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Friday, 11 July 2008
Genetic Recombination gets More Precise
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Genes That Control Embryonic Stem Cell Fate Identified
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Stem Cells Restore Muscle in Muscular Dystrophy
 Researchers at the Joslin Diabetes Center have demonstrated for the first time that transplanted muscle stem cells can both improve muscle function in animals with a form of muscular dystrophy and replenish the stem cell population for use in the repair of future muscle injuries.
By injecting purified stem cells isolated from adult skeletal muscle, researchers have shown they can restore healthy muscle and improve muscle function in mice with a form of muscular dystrophy. Those muscle-building stem cells were derived from a larger pool of so-called satellite cells that normally associate with mature muscle fibers and play a role in muscle growth and repair.
In addition to their contributions to mature muscle, the injected cells also replenished the pool of regenerative cells normally found in muscle. Those stem cells allowed the treated muscle to undergo subsequent rounds of injury repair, they found.
"I'm very excited about this," said lead author Amy J. Wagers, Ph.D., Principal Investigator in the Joslin Section on Developmental and Stem Cell Biology, principal faculty member at the Harvard Stem Cell Institute and Assistant Professor of Stem Cell and Regenerative Biology at Harvard University.
"This study indicates the presence of renewing muscle stem cells in adult skeletal muscle and demonstrates the potential benefit of stem cell therapy for the treatment of muscle degenerative diseases such as muscular dystrophy."
"Our work shows proof-of-concept that purified muscle stem cells can be used in therapy," said Wagers, noting that in some cases the stem cells replaced more than 90 percent of the muscle fibers. Such an advance would require isolation of stem cells equivalent to those in the mouse from human muscle, something Wagers said her team is now working on.
The study was designed to test the concept that skeletal muscle precursor cells could function as adult stem cells and that transplantation of these cells could both repair muscle tissue and regenerate the stem cell pool in a model of Duchenne muscular dystrophy, she said.
Duchenne muscular dystrophy is the most common form of the disease and is characterized by rapidly progressing muscle degeneration. The disease is caused by a genetic mutation and there is currently no cure.
The data from this new study demonstrate that regenerative muscle stem cells can be distinguished from other cells in the muscle by unique protein markers present on their surfaces. The authors used these markers to select stem cells from normal adult muscle and transferred the cells to diseased muscle of mice carrying a mutation in the same gene affected in human Duchenne muscular dystrophy.
Satellite cells were first described decades ago and have since generally been considered as a homogeneous group, Wagers said. While anatomically they look similar under a microscope, they nonetheless show considerable variation in their physiology and function. In a previous study, Wagers' identified a set of five markers that characterize the only subset of satellite cells responsible for forming muscle, which they also refer to as skeletal muscle precursors or SMPs.
In the new study, the researchers analyzed the stem cell and regenerative properties of those SMPs. When engrafted into muscle of mice lacking dystrophin, purified SMPs contributed to up to 94 percent of muscle fibers, restoring dystrophin expression and significantly improving muscle structure and contractile function, they report. (The dystrophin gene encodes a protein important for muscle integrity. Mice lacking dystrophin, also known as mdx mice, are a model for Duchenne muscular dystrophy, the most prevalent form of muscular dystrophy.)
"Importantly, high-level engraftment of transplanted SMPs in mdx animals shows therapeutic value — restoring defective dystrophin gene expression, improving muscle histology, and rescuing physiological muscle function," the researchers said.
"Moreover, in addition to generating mature muscle fibers, transplanted SMPs also re-seed the satellite cell niche and are maintained there such that they can be recruited to participate in future rounds of muscle regeneration.”
"Taken together, these data indicate that SMPs act as renewable, transplantable stem cells for adult skeletal muscle. The level of myofiber reconstitution achieved by these myogenic stem cells exceeds that reported for most other myogenic cell populations and leads to a striking improvement of muscle contraction function in SMP-treated muscles. These data thus provide direct evidence that prospectively isolatable, lineage-specific skeletal muscle stem cells provide a robust source of muscle replacement cells and a viable therapeutic option for the treatment of muscle degenerative disorders."
Wagers noted however that there may be complications in the delivery of cell therapy in humans, particularly for those with conditions influencing skeletal muscle throughout the body. Even so, the new findings present an "opportunity to understand what happens [to these regenerative cells] in disease and identify factors and pathways that may boost their activity," she said.
"We may get a handle on drugs that could target muscle impairment" not only in those with muscular dystrophies, but also in elderly people suffering from the muscle wasting that comes with age.
"Once the healthy stem cells were transplanted into the muscles of the mice with muscular dystrophy, they generated cells that incorporated into the diseased muscle and substantially improved the ability of the treated muscles to contract," said Wagers.
"At the same time, the transplantation of the healthy stem cells replenished the formerly diseased stem cell pool, providing a reservoir of healthy stem cells that could be re-activated to repair the muscle again during a second injury."
According to the paper, these cells provide an effective source of immediately available muscle regenerative cells as well as a reserve pool that can maintain muscle regenerative activity in response to future challenges.
"This work demonstrates, in concept, that stem cell therapy could be beneficial for degenerative muscle diseases," Wagers said.
Wagers also said the study will lead to other studies in the near-term that will identify pathways that regulate these muscle stem cells in order to figure out ways to boost the normal regenerative potential of these cells. These could include drug therapies or genomic approaches, she said. In the long-term, the idea will be to replicate these findings in humans.
"This is still very basic science, but I think we're going to be able to move forward in a lot of directions. It opens up many exciting avenues," she said.
The Wagers Lab at Joslin studies both hematopoietic stem cells, which constantly maintain and can fully regenerate the entire blood system, as well as skeletal muscle stem cells, involved in skeletal muscle growth and repair. The work is aimed particularly at defining novel mechanisms that regulate the migration, expansion, and regenerative potential of these two distinct adult stem cells.
About Joslin Diabetes Center
Joslin Diabetes Center is the world's largest diabetes clinic, diabetes research center and provider of diabetes education. Joslin is dedicated to ensuring people with diabetes live long, healthy lives and offers real hope and progress toward diabetes prevention and a cure for the disease. Founded in 1898 by Elliott P. Joslin, M.D., Joslin is an independent non-profit institution affiliated with Harvard Medical School.
Reference:
Highly Efficient, Functional Engraftment of Skeletal Muscle Stem Cells in Dystrophic Muscles
Massimiliano Cerletti, Sara Jurga, Carol A. Witczak, Michael F. Hirshman, Jennifer L. Shadrach, Laurie J. Goodyear, and Amy J. Wagers
Cell, Vol 134, 37-47, 11 July 2008
.........
ZenMaster
Researchers at the Joslin Diabetes Center have demonstrated for the first time that transplanted muscle stem cells can both improve muscle function in animals with a form of muscular dystrophy and replenish the stem cell population for use in the repair of future muscle injuries.
By injecting purified stem cells isolated from adult skeletal muscle, researchers have shown they can restore healthy muscle and improve muscle function in mice with a form of muscular dystrophy. Those muscle-building stem cells were derived from a larger pool of so-called satellite cells that normally associate with mature muscle fibers and play a role in muscle growth and repair.
In addition to their contributions to mature muscle, the injected cells also replenished the pool of regenerative cells normally found in muscle. Those stem cells allowed the treated muscle to undergo subsequent rounds of injury repair, they found.
"I'm very excited about this," said lead author Amy J. Wagers, Ph.D., Principal Investigator in the Joslin Section on Developmental and Stem Cell Biology, principal faculty member at the Harvard Stem Cell Institute and Assistant Professor of Stem Cell and Regenerative Biology at Harvard University.
"This study indicates the presence of renewing muscle stem cells in adult skeletal muscle and demonstrates the potential benefit of stem cell therapy for the treatment of muscle degenerative diseases such as muscular dystrophy."
"Our work shows proof-of-concept that purified muscle stem cells can be used in therapy," said Wagers, noting that in some cases the stem cells replaced more than 90 percent of the muscle fibers. Such an advance would require isolation of stem cells equivalent to those in the mouse from human muscle, something Wagers said her team is now working on.
The study was designed to test the concept that skeletal muscle precursor cells could function as adult stem cells and that transplantation of these cells could both repair muscle tissue and regenerate the stem cell pool in a model of Duchenne muscular dystrophy, she said.
Duchenne muscular dystrophy is the most common form of the disease and is characterized by rapidly progressing muscle degeneration. The disease is caused by a genetic mutation and there is currently no cure.
The data from this new study demonstrate that regenerative muscle stem cells can be distinguished from other cells in the muscle by unique protein markers present on their surfaces. The authors used these markers to select stem cells from normal adult muscle and transferred the cells to diseased muscle of mice carrying a mutation in the same gene affected in human Duchenne muscular dystrophy.
Satellite cells were first described decades ago and have since generally been considered as a homogeneous group, Wagers said. While anatomically they look similar under a microscope, they nonetheless show considerable variation in their physiology and function. In a previous study, Wagers' identified a set of five markers that characterize the only subset of satellite cells responsible for forming muscle, which they also refer to as skeletal muscle precursors or SMPs.
In the new study, the researchers analyzed the stem cell and regenerative properties of those SMPs. When engrafted into muscle of mice lacking dystrophin, purified SMPs contributed to up to 94 percent of muscle fibers, restoring dystrophin expression and significantly improving muscle structure and contractile function, they report. (The dystrophin gene encodes a protein important for muscle integrity. Mice lacking dystrophin, also known as mdx mice, are a model for Duchenne muscular dystrophy, the most prevalent form of muscular dystrophy.)
"Importantly, high-level engraftment of transplanted SMPs in mdx animals shows therapeutic value — restoring defective dystrophin gene expression, improving muscle histology, and rescuing physiological muscle function," the researchers said.
"Moreover, in addition to generating mature muscle fibers, transplanted SMPs also re-seed the satellite cell niche and are maintained there such that they can be recruited to participate in future rounds of muscle regeneration.”
"Taken together, these data indicate that SMPs act as renewable, transplantable stem cells for adult skeletal muscle. The level of myofiber reconstitution achieved by these myogenic stem cells exceeds that reported for most other myogenic cell populations and leads to a striking improvement of muscle contraction function in SMP-treated muscles. These data thus provide direct evidence that prospectively isolatable, lineage-specific skeletal muscle stem cells provide a robust source of muscle replacement cells and a viable therapeutic option for the treatment of muscle degenerative disorders."
Wagers noted however that there may be complications in the delivery of cell therapy in humans, particularly for those with conditions influencing skeletal muscle throughout the body. Even so, the new findings present an "opportunity to understand what happens [to these regenerative cells] in disease and identify factors and pathways that may boost their activity," she said.
"We may get a handle on drugs that could target muscle impairment" not only in those with muscular dystrophies, but also in elderly people suffering from the muscle wasting that comes with age.
"Once the healthy stem cells were transplanted into the muscles of the mice with muscular dystrophy, they generated cells that incorporated into the diseased muscle and substantially improved the ability of the treated muscles to contract," said Wagers.
"At the same time, the transplantation of the healthy stem cells replenished the formerly diseased stem cell pool, providing a reservoir of healthy stem cells that could be re-activated to repair the muscle again during a second injury."
According to the paper, these cells provide an effective source of immediately available muscle regenerative cells as well as a reserve pool that can maintain muscle regenerative activity in response to future challenges.
"This work demonstrates, in concept, that stem cell therapy could be beneficial for degenerative muscle diseases," Wagers said.
Wagers also said the study will lead to other studies in the near-term that will identify pathways that regulate these muscle stem cells in order to figure out ways to boost the normal regenerative potential of these cells. These could include drug therapies or genomic approaches, she said. In the long-term, the idea will be to replicate these findings in humans.
"This is still very basic science, but I think we're going to be able to move forward in a lot of directions. It opens up many exciting avenues," she said.
The Wagers Lab at Joslin studies both hematopoietic stem cells, which constantly maintain and can fully regenerate the entire blood system, as well as skeletal muscle stem cells, involved in skeletal muscle growth and repair. The work is aimed particularly at defining novel mechanisms that regulate the migration, expansion, and regenerative potential of these two distinct adult stem cells.
About Joslin Diabetes Center
Joslin Diabetes Center is the world's largest diabetes clinic, diabetes research center and provider of diabetes education. Joslin is dedicated to ensuring people with diabetes live long, healthy lives and offers real hope and progress toward diabetes prevention and a cure for the disease. Founded in 1898 by Elliott P. Joslin, M.D., Joslin is an independent non-profit institution affiliated with Harvard Medical School.
Reference:
Highly Efficient, Functional Engraftment of Skeletal Muscle Stem Cells in Dystrophic Muscles
Massimiliano Cerletti, Sara Jurga, Carol A. Witczak, Michael F. Hirshman, Jennifer L. Shadrach, Laurie J. Goodyear, and Amy J. Wagers
Cell, Vol 134, 37-47, 11 July 2008
.........
ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Primer on Mitochondria

1. What are mitochondria and where are they found?
Mitochondria are bean-shaped compartments within cells that supply energy. These compartments, a type of membrane-bound organelle, are found in eukaryotes — organisms whose cells have nuclei, the home of the genome. Multicellular organisms (humans, mice, fish, etc.) as well as some unicellular ones, like yeast, are counted as eukaryotes. Bacteria, though, are not: They are considered prokaryotes for their lack of organelles, including mitochondria and nuclei. Intriguingly, mitochondria vary widely across organisms and even within an organism. Drastic differences can exist in the number of mitochondria per cell, their size and morphology, and even their biochemical capabilities. For example, fatty acids readily broken down by mitochondria in muscle, but not brain tissue. Because of a lack of molecular knowledge about mitochondria and their resident proteins, the basis for such differences is largely unclear.
2. What do mitochondria do? Although mitochondria are perhaps best known for their roles in energy metabolism, they also participate in a plethora of other key biological processes. These include critical functions such as programmed cell death (or “apoptosis”), a normal mechanism through which old or damaged cells can be eliminated. Defects in mitochondria are associated with more than 50 human diseases, ranging from in-born errors of metabolism in infants to neurodegeneration in adults. Moreover, several common diseases, such as cancer and type 2 diabetes, have been associated with mitochondrial dysfunction. Prescription drugs can also disrupt mitochondria. Such drug-induced toxicity is a reason why some drugs are pulled from the market and why some potential drugs fail the clinical trial process.
3. Where do mitochondria come from? Mitochondria, it turns out, have their own tiny genome. And in humans, this mitochondrial DNA is inherited solely from the mother. Such maternal inheritance arises because mitochondria from sperm are lost following fertilization, while those contributed by the egg persist. Because it is maternally inherited, mitochondrial DNA can provide clues about human history, including the most recent common matrilineal ancestor of living humans (so-called “Mitochondrial Eve”.) But there are, in fact, paternal contributions to mitochondria. The parts of the mitochondria that are derived from nuclear genes actually come from both parents (see below). This follows a core principle of human genetics: of the 23 pairs of chromosomes that make up your nuclear genome, roughly half come from Mom and the other half from Dad. Evolutionarily speaking, mitochondria have a very interesting history. They are descendants of an ancient bacterium — a relative of the modern bacterial species, Rickettsia prowazekii — that some 2 billion years ago was enveloped by another cell. That moment marked the beginning of a long and mutually beneficial relationship with eukaryotic cells, known as endosymbiosis. As a result of such “co-habitation”, eukaryotic cells and mitochondria have evolved and adapted to life together, such that now, neither can survive alone.
4. Where do the proteins in mitochondria come from? Because of the organelle’s unusual past, the molecular pieces that make up mitochondria have undergone some shuffling of their own. Mitochondria carry a small circular genome, a vestige of their days as free-living bacteria that has been winnowed during evolution to just a few protein-coding genes. The human mitochondrial genome was decoded in 1981, a full 20 years before the human genome itself was decoded. The organelle’s genome consists of roughly 16,000 chemical units called base pairs, much smaller than the nuclear genome’s 3 billion base pairs. The mitochondrial genome includes just 37 genes: 13 genes that encode proteins and 24 additional non-protein coding genes. The rest of the genes required for a functioning mitochondrion, roughly 1,200 to 1,500 in total, now reside in the nucleus. Identifying these genes from DNA sequence data alone has proven immensely difficult, which is why other large-scale approaches — namely proteomics and computational methods — are required to pinpoint them.
See also:
MitoCarta - Protein Catalogue for Mitochondria
CellNEWS - Friday, 11 July 2008
.........
ZenMaster
For more on stem cells and cloning, go to CellNEWS
at http://cellnews-blog.blogspot.com/

